Center for Clinical Research (CCR)

The ACTRI Center for Clinical Research is under the co-direction of Mark S. Wallace, MD and Kathryn Gold, MD. The Center encompass services that facilitate performing all aspects of clinical research from Idea to Innovation.
More than two dozen employees – skilled nurses, a registered dietitian, a registered sonographer, clinical coordinators, project managers, pharmacists, and pharmacy technicians – support the researchers who conduct clinical trials at the ACTRI.
The ACTRI Clinics

The ACTRI Clinics operate across two locations to support diverse clinical research needs. The primary site, ACTRI@La Jolla, is located at UC San Diego’s Medical Campus in La Jolla. It spans 18,000 square feet within the ACTRI building and offers comprehensive clinical research services, including clinical space, a Phase I unit, nursing, pharmacy, and research coordination. This facility currently supports over 350 clinical trials including pediatric and adult, advancing groundbreaking research across multiple disciplines.
The ACTRI@Linda Vista Center for Clinical Research focuses on pediatric clinical research, providing dedicated clinical space, a neurocognitive testing room, lab processing, and nursing services. Together, these locations enhance ACTRI’s ability to facilitate high-quality, patient-centered research, ensuring access to state-of-the-art resources for investigators and participants alike.
Research Pharmacy

Pharmacy services are critical to the success of clinical trials involving experimental drugs.
The ACTRI pharmacist is an active member of the research team, helping with the design of the study and participating in protocol review.
The Pharmacy team is responsible for:
Procuring and storing drugs;
- Maintaining all records regarding their purchase, regulation, and dispensing;
- Compounding, labeling and dispensing study drugs;
- Keeping accountability records;
- Assuring quality control and compliancy with federal, state, and institutional guidelines.
The ACTRI Research Pharmacy is located within the ACTRI@La Jolla clinic, which better serves our investigators, clinic staff, and subjects by eliminating wait times, drug stability concerns and transport responsibilities, therefore improving general clinical trial safety.
For more information, e-mail Ji Sun, PharmD, PhD, Investigational Drug Service Manager.
Please note the links to the Recharge Rates are for UC San Diego researchers using their @health.ucsd.edu (NOT @ucsd.edu) AD credentials. Non UCSD Researchers or for other inquiries, use the Contacts listed above.
MRI Research Center
The MRI Research Center includes a state-of-the-art 3T MRI scanner that offers high-resolution structural imaging, as well as dynamic imaging techniques.
It can be used for a wide range of studies, including Alzheimer’s disease, liver disease, and musculoskeletal disease.
L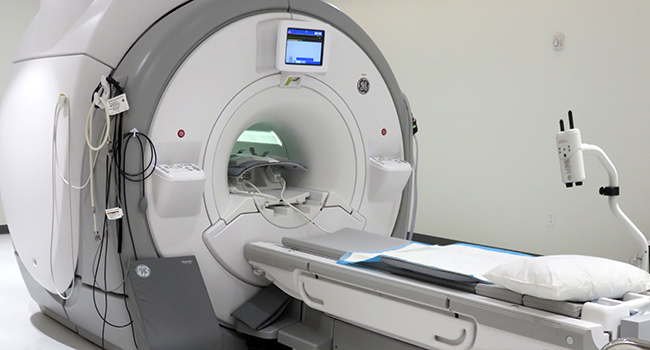 ocated in a lower level suite of the ACTRI building, the MRI Research Center is a shared resource of the ACTRI Centers for Clinical Research and Radiology Department at UC San Diego and is operated by the Center for Translational Imaging for Precision Medicine (CTIPM).
ocated in a lower level suite of the ACTRI building, the MRI Research Center is a shared resource of the ACTRI Centers for Clinical Research and Radiology Department at UC San Diego and is operated by the Center for Translational Imaging for Precision Medicine (CTIPM).
Research subjects will check in at the ACTRI@La Jolla clinic on the first floor and are then escorted to the MRI Research Center.
For technical questions about the MRI Research Center and its capabilities, contact Jenna Vuong.
MRS Research Center Recharge Rates
Please note the links to the Recharge Rates are for UC San Diego researchers using their @health.ucsd.edu (NOT @ucsd.edu) AD credentials. Non UCSD Researchers or for other inquiries, use the Contacts listed above.
Clinical Research Coordinators
Clinical Trial Coordination
The staffing needed for a clinical research project is determined by the kind of study being conducted. Although the project's budget must be kept in mind, adequate staffing is critical to the safety and success of the study.
While the principal investigator is ultimately responsible for the conduct of a study, including the safety and welfare of any clinical research participants, tasks such as the following are commonly delegated to the ACTRI Clinical Research Coordinators:
-
Overseeing screening and enrollment
-
Conducting the informed consent process
-
Documenting eligibility of study participants
-
Conducting study visits
- Obtaining vital signs, ECGs
-
Obtaining and shipping specimens
-
Completing case report forms
-
Preparing adverse event forms
-
Developing quality assurance and quality control measures
-
Communicating with the study sponsor
Coordinator services are provided on an hourly basis or can be assigned at a percent effort to an individual investigator for a specified period of time. Coordinators are available to assist on a study for a maximum of 50 percent time.
Language Translation Services for Consent Forms and Other Documentation
The ACTRI Coordinators can provide English to Spanish Translation services for ACTRI members.
For other languages, ACTRI members can contact Language Translation, Inc Services at info@languagetranslation.com or call (619) 516-4037. Translation certification is available for IRB and regulatory submissions.
For questions about Clinical Research Coordinator services, please contact us at ACTRI-coordservices@health.ucsd.edu.
Clinical Research Coordinators Recharge Rates
Please note the links to the Recharge Rates are for UC San Diego researchers using their @health.ucsd.edu (NOT @ucsd.edu) AD credentials. Non UCSD Researchers or for other inquiries, use the Contacts listed above.
Clinical Trial Support Services: Regulatory Affairs and Regulatory Support
“The CTSS team has been the backbone of our early research success. Their knowledge, attention to detail, and unwavering support made it possible to navigate complex regulatory hurdles and bring our studies to life.”
--- Dr. Gabriel Schnickel, Surgical Director, Liver Transplantation, Division of Transplant & Hepatobiliary Surgery
“Doing clinical trials as a clinically practicing physician is difficult. There are a lot of regulatory, budgetary, and safety coordination that must be carefully coordinated. The CTSS at ACTRI made the process relatively smooth and efficient ensuring that we had all the appropriate documentation in place, providing adequate research training to our staff, and helping push towards an expedited trial opening. These important clinical trials could not be done without their assistance.”
-- Dr. Zachary T. Berman, Associate Professor of Clinical Radiology, Division of Vascular and Interventional Radiology
The Clinical Trial Support Service (CTSS) team interact closely with UCSD investigators to provide quality services that support the planning, implementation, and completion of the clinical trials and clinical research studies.
Regulatory affairs
Our experienced staff can provide the following services:
- Determination of product classification
- FDA IND/IDE/510K Guidance and Support
- Protocol development assistance
- Assistance with IND/IDE applications
Regulatory support
The CTSS has staff with regulatory experience who can assist researchers with various IRB and ancillary committee submissions.
Services include:
- Prepare IRB submissions including for non-UCSD IRBs – New applications, amendments, renewals, and reportable events
- Informed Consent development
- Coordination with Ancillary Committees – including Conflict of Interest, Human Exposure Review, and Institutional Biosafety
- Work with Sponsors and Clinical Research Organizations
- Partner with ACTRI Research Coordinators
For more detailed information or to discuss how we can assist you, please contact us at actri-ctss@health.ucsd.edu
Additional UCSD resources are available regarding the Office of Compliance and Privacy; Research Compliance and Integrity; Research Ethics, and the Office of IRB Administration (OIA)
Clinical Trial Support Services Recharge Rates
Please note the links to the Recharge Rates are for UC San Diego researchers using their @health.ucsd.edu (NOT @ucsd.edu) AD credentials. Non UCSD Researchers or for other inquiries, use the Contacts listed above.
Clinical Trial Support Services: Project and Data Management
To increase the speed and efficiency of clinical research protocols, the ACTRI is offering project management and support services to investigators. ACTRI can help launch and manage your studies by facilitating the initiation and execution of clinical protocols, enabling you to focus on your research.
Applied to clinical trials, project management is critical to ensuring that trials are set up, enrolled, conducted, and reported on-time and according to budget.
The ACTRI Clinical Trials Project Manager (PM)
- Coordination of multi-site research
- Assist in preparation of annual reports
- Clinical Trial Feasibility Assessment
- Site Qualification Preparation and Support
- Collaboration with Office of Clinical Trial Agreements and Office of Coverage Analysis Administration via Kuali Research
- Study Start-up Coordination
- Serve as a key point of contact for research studies, working with sponsors and CROs
- Develop and maintain study metrics
- Review, triage and track resources and service requests across ACTRI and UCSD research units;
- Develop reports/tools to track enrollment using low-code/no-code software capabilities
- Create data capture processes/systems
Clinical Trial Invoicing
The Clinical Trial Support Service (CTSS) team works closely with investigators and research teams who require additional support to invoice industry sponsors of clinical trials. This service streamlines the financial management of industry sponsored clinical trials by managing invoicing, tracking payment, and providing financial oversight from study activation to closeout.
Service includes:
- Review the fully executed Clinical Trial Agreement (CTA) and collaborate with study teams to identify invoiceable items
- Create invoice and review financial report to confirm check is deposited
- Review and claim unapplied/unidentified checks
- Assist with billing dispute resolution in collaboration with Accounts Receivable team
- Reconcile study billing account and create final invoice at study closeout
Note: Priority will be given to investigators currently using ACTRI services.
Investigators are encouraged to submit a request for clinical trial invoicing during study start up. However, retrospective financial review of a study will be considered on a case by case basis.
For more detailed information or to discuss how we can assist you, please contact us at actri-ctss@health.ucsd.edu.
Clinical Trials Support Services Recharge Rates
For more detailed information or to discuss how we can assist you, please contact us at actri-ctss@health.ucsd.edu
Clinical Trial Support Services Recharge Rates
Please note the links to the Recharge Rates are for UC San Diego researchers using their @health.ucsd.edu (NOT @ucsd.edu) AD credentials. Non UCSD Researchers or for other inquiries, use the Contacts listed above.
Multi-Network Consortium
The Trial Innovation Network (TIN)
TIN is a collaborative initiative to address critical roadblocks in clinical trials and accelerate the translation of novel interventions into life-saving therapies. It was launched to facilitate multi-site clinical studies.
The network focuses on operational innovation, operational excellence, and collaboration while leveraging the expertise and resources of the CTSA Program.
Features include:
- Single IRB reliance model system – Flexible and efficient IRB review for multicenter studies:
- SMART IRB Exchange – The Streamlined, Multisite, Accelerated Resources for Trials (SMART) IRB Exchange is an IT platform, supported by the Duke/Vanderbilt TIC, used to initiate or activate IRB reliance for a given study, as well as to document ongoing IRB approvals for the study, promote transparency, and facilitate communication. It is used to support single IRB review of all Network studies.
- Master contracting agreements
- Streamlined protocols – Master protocol and clinical research form (CRF) templates
- Quality by design approaches
- Focus on evidence-based strategies to recruitment and patient engagement (Harmonized IT tools and approaches, Data-driven recruitment strategies)
The key organizational partners:
- The CTSA Program Hub
- The Trial Innovation Centers (TICs):
- focused on operational excellence, operational innovation, and quality by design
- charged with coordinating and providing innovative, high quality operational support for clinical trials
- The Recruitment Innovation Center (RIC):
- focused on innovative and evidence-based approaches to participant recruitment, retention, and engagement
- provide tools and services to enhance participant recruitment and retention
- Trial Innovation Network Hub Liaison Teams
- Frontline of the Trial Innovation Network, will lead the Network at the local level and will connect the Hubs to the TICs and RIC.
- will use their experience and knowledge of the local environment to coordinate Trial Innovation activities at their Institutions, tailoring general Network plans into more specific action plans that are best suited for their Hubs.
- UCSD Altman Clinical and Translational Research Institute is a TIN Hub Liaison Team.
Proposal Intake Process
- The Trial Innovation Network is now accepting Network Project Proposals. Visit the Trial Innovation Network website to submit a Network Project Proposal.
- Level 1 Network Projects (Basic Consultation or Service)
- The TIN provides specific consultations or services (up to a pre-specified threshold) but is not responsible for recruiting patients or managing study data.
- Level 2 Network Projects (Comprehensive Consultation)
- The TIN provides comprehensive in-kind consultations (up to a pre-specified threshold) to develop a proposal into a protocol.
- A Comprehensive Consultation could include: in-depth protocol development, statistics, recruitment feasibility, recruitment plans, study budgets, and other key elements. During the Comprehensive Consultation, input from the CTSA Program Hubs on the protocol and proposed study budgets would be sought.
- After a Level 2 Network Project Comprehensive Consultation, the Principal Investigator has the option to independently develop an application for a clinical trial using the TICs/RIC as the Coordinating Centers and the Hubs as the recruitment sites.
Please note: Prior to submitting a proposal to the NIH, UCDS investigators are required to contact the ACTRI Hub Liaison Team to discuss their proposal and submit it for a pre-review by the ACTRI Scientific Review Committee.
Please contact ACTRI team medical director Mark S. Wallace or team project manager Archana Bhatt to begin the consultation and pre-review process.
UC BRAID Network
The University of California Biomedical Research Acceleration, Integration, and Development (UC BRAID) is a consortium of University of California health campuses - Davis, Irvine, Los Angeles, San Diego, and San Francisco, each of which is a recipient of an NIH Clinical and Translational Science Award.
Building on the combined strengths of the individual campus, UC BRAID aims to accelerate research and improve health through collaboration, resource sharing and infrastructure development. We aim to catalyze, accelerate, and reduce the barriers for biomedical, clinical, and translational research across the UC system. The effort includes identifying shared challenges and developing system-wide solutions.
Areas of continued attention include industry contracting, institutional review boards (IRBs), and participant recruitment. UC BRAID's early successes in sharing data and infrastructure were demonstrated in the cohort identification tool UC ReX, which now serves as a model for the shared benefit of a nationwide collaboration.
Meet the CCR staff
Clinic Nurses

Bernadette Cale, MSN, RN
Assistant Clinical Director
 |
 |
 |
 |
| Patricia Santana, RN Assistant Clinical Manager |
Jessica Webb, RN Clinical Nurse |
Clinical Nurse
|
Emily Rizo, NP Clinical Nurse |
|
|
|
 |
 |
|
Clinical Nurse
|
Clinical Nurse
|
Clinical Nurse
|
Clinical Nurse
|
 |
 |
||
| Chandelle Butterfield, RN
Clinical Nurse
|
Asami Mimura, LVN Clinical Nurse
|
Clinic Lab, Nutrition, Sonography, and Administrative Staff
|
|
 |
 |
 |
| Macarena Lomeli "Nina" Administrative Coordinator |
Crystal Groom Lab Manager |
Dietitian
|
Sonographer
|
|
|
|
 |
 |
| Nelson Guanipa Lab technician |
Operations Specialist
|
Jesse Lora Operations Specialist |
Dionne Robinson Lab technician |
Pharmacy Staff
 |
|||
|
Pharmacy Manager
|
|||
|
|
|
 |
 |
| Marianne Tutto IDS Pharmacist |
Thaine Ross |
Brendan Costello |
Julian Leland IDS Pharmacist |
Clinical Trial Support Team
|
|
 |
|
| Archana Bhatt Program Manager, CTSS |
Lea Jacinto Regulatory Project Manager |
Regulatory Project Manager
|
 |
||
| Kristina Laws Regulatory Project Manager |
Clinical Research Coordinators
 |
|
 |
 |
| Fanny Delebecque Clinical Research Program Manager |
Deyna Arellano Clinical Research Coordinator |
Katherine Bunda Clinical Research Coordinator |
Gayle Dizon Clinical Research Coordinator |
|
|
 |
 |
|
|
Clinical Research Coordinator
|
Rosalee Espiritu Assistant Clinical Research Coordinator | Jazmin Morales Assistant Clinical Research Coordinator |
Nicolle Mendez Clinical Research Coordinator |







